Adapting to low oxygen
A Biochemical Study of Comb Jellies
A Biochemical Approach to Ctenophore Survival in Anoxia
Kristin D. Henderson & Erik V. Thuesen
PROJECT BACKGROUND
Ctenophores, jellyfish-like animals that thrive in low oxygen environments, are thought to do so because of metabolic adaptations that allow them to take oxygen from very low oxygen concentrations. As a comparison, this would be analogous to humans being able to cope well in low oxygen environments, such as at high altitudes. Puget Sound ctenophores (e.g. Pleurobrachia bachei) can survive for extended periods without oxygen, but Mnemiopsis leidyi, a species native to Chesapeake Bay, has no tolerance to anoxia. This study investigates the biochemical differences between ctenophores in Puget Sound, Hood Canal, Chesapeake Bay and deep-sea oxygen minimum layers. Oxygen depletion may have an enormous effect on the abundances, diversity, and interactions of marine organisms (Keister et al., 2000; Diaz and Rosenburg, 1995) by concentrating less hypoxia tolerant organisms in oxygenated waters. In the Chesapeake, M. leidyi population sizes are increasing with increasing environmental degradation and resulting low oxygen levels, and they have begun to outcompete fish larvae. These ctenophores may also impact the shellfish and crab industries. The ultimate goal of this project is to help unravel the mechanisms that zooplankton use to live in low oxygen estuaries to better understand the impacts of worsening environmental conditions in Puget Sound.
CTENOPHORES
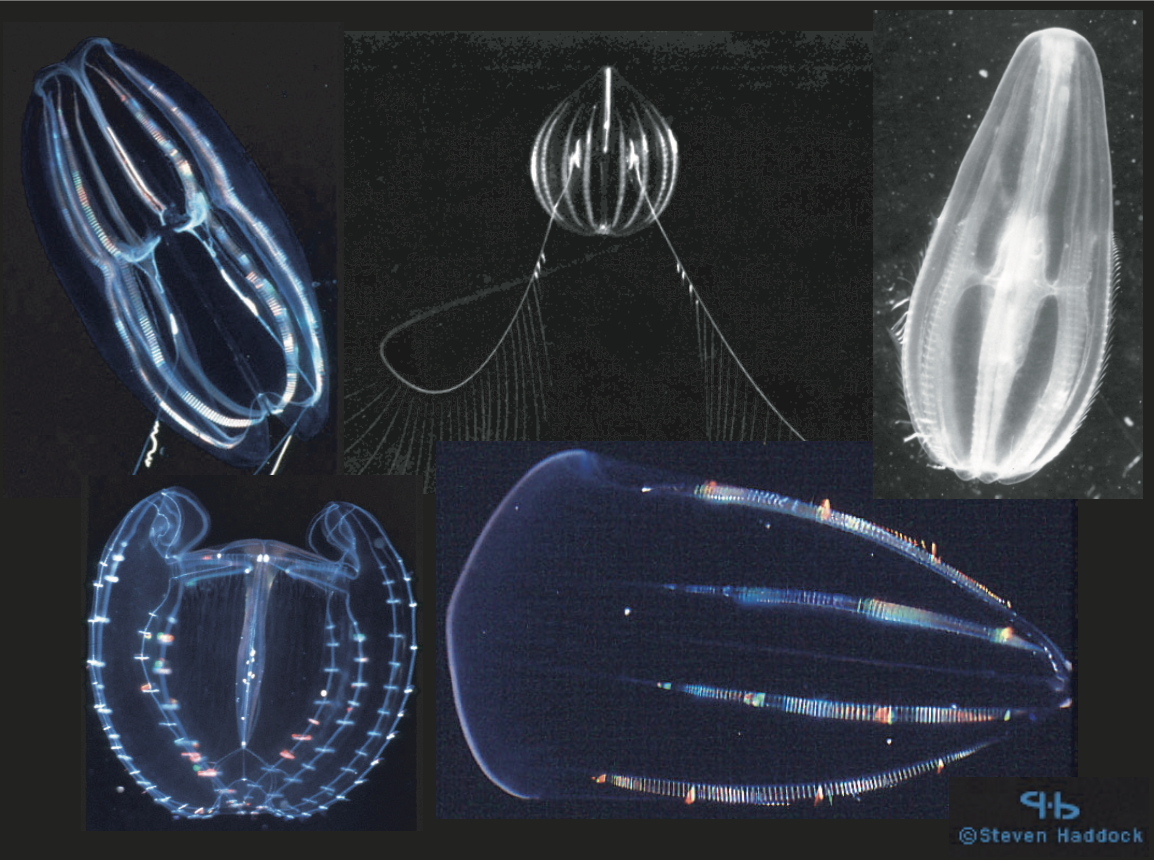
Ctenophores are gelatinous zooplankton (Fig. 1) similar to jellyfish in that they are carnivorous and are often transparent. They live only in marine environments ranging from estuarine to oceanic, surface to deep-sea, and polar to equatorial. Ctenophores are characterized by eight comb rows of ciliated plates or ctenes which are used for locomotion. Our study includes species representing three different ctenophore Orders (i.e. Cydippida, Lobata, Beroida), each characterized by different means of locomotion and feeding strategies (Wrobel and Mills, 1998).
HYPOXIA
Low dissolved oxygen concentrations occur as episodic events in estuarine environments and are persistent in the oxygen minimum layers of the deep sea (~700 meters). Hypoxia generally occurs in estuaries in summer when density stratification of the water column occurs. Oxygen concentrations decrease from over-enrichment of nutrients, such as nitrogen from fertilizer and sewage runoff. Anoxia is a complete lack of oxygen.
ECOPHYSIOLOGY OF ANIMALS IN HYPOXIA
Possible Responses to Hypoxia
- Swim away
- Regulate oxygen uptake by aerobic means
- Use anaerobic energy
- Go into metabolic arrest (similar to hibernation)
- Die
ORGANISM COLLECTION
Pleurobrachia bachei and Nephalotena sp. were hand dipped from southern Puget Sound and Friday Harbor (Fig. 2). Specimens were transported to the laboratory and maintained at 10°C in 30 psu filtered seawater (FSW). Beroe spp. were hand collected by scuba divers (Fig. 2). Hormiphora californensis were collected by suction sampler device on the Monterey Bay Aquarium Research Institute's ROV Tiburon April 5-11, 2005 (Fig. 3). Specimens were held without food for 24 h when possible or until their guts were empty, before being frozen and stored in liquid nitrogen until being used for enzyme assay investigations.
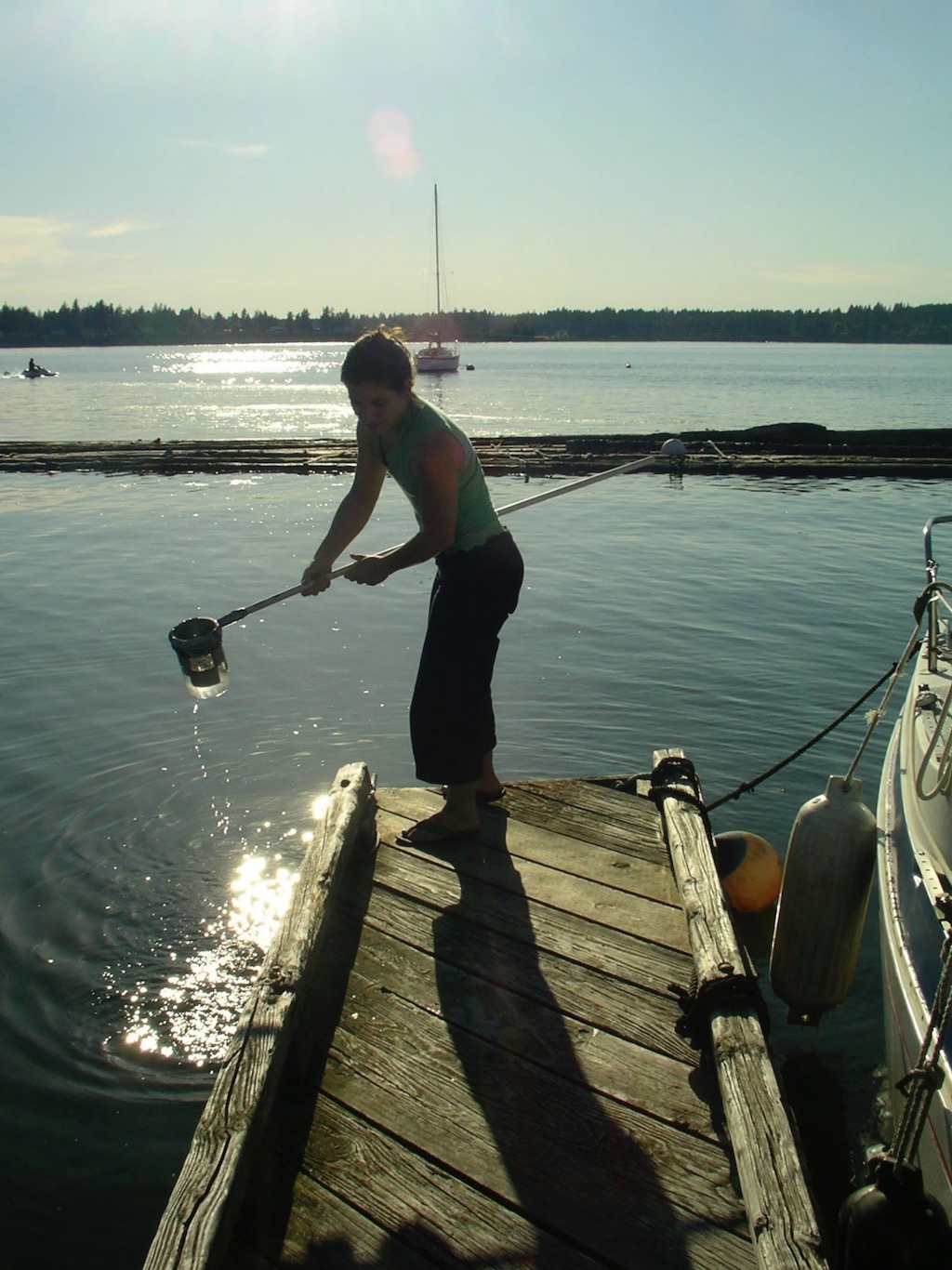

ENZYMES AS INDICATORS OF METABOLIC ACTIVITY
Citrate synthase (CS), an important enzyme in the citric acid cycle acts as an indicator of aerobic metabolic potential. Two glycolytic enzymes, lactate dehydrogenase (LDH) and octopine dehydrogenase (ODH), act as indicators of anaerobic metabolic potential. Amounts of CS activity relative to LDH or ODH activity indicate the importance and role of these aerobic or anaerobic pathways in coping with hypoxia. If CS activity is greater in organisms with greater ctene numbers, and the mitochondria associated with them, ctenophores with a greater number of ctenes may tolerate hypoxia by this aerobic adaptation. Ctenophores do not rely on burst swimming capacity but on their transparency to avoid predators, therefore they do not use anaerobic energy for burst swimming. Greater levels of LDH or ODH may indicate an anaerobic adaptation to hypoxia.
ENZYME ASSAYS
Whole ctenophores were weighed on an analytical balance while still frozen and homogenized with 1:1 parts weight to volume of 50 mM imidazole/HCl homogenization buffer using Duall hand held glass homogenizers kept on ice. The homogenates were centrifuged at 6000 x g for 10 min at 2°C. All assays were performed within 1 h of homogenization using a Hewlett-Packard diode array spectrophotometer equipped with a water-jacketed cuvette holder. Measurements of enzyme activity were made in 2 ml quartz cuvettes at 20°C under non-limiting conditions in order to estimate maximum metabolic potential following procedures described previously (Childress and Somero, 1979; Somero and Childress, 1980). Activity measurements of CS were performed in a medium containing 50 mM imidazole/HCl buffer (pH 7.8 at 20°C), 1 mM oxaloacetate, 0.1 mM acetyl-CoA, 0.2 mM 5, 5-dithiobis (2-nitrobenzoic acid) (DTNB), and 1.5 mM MgCl2. The increase in absorbance at 412 nm due to the reaction of the reduced coenzyme A generated by the enzymatic reaction with DTNB was recorded. We recorded background activity after the addition of homogenate supernatant, and this background rate was subtracted from the overall rate after the assay reaction was initiated by addition of the oxaloacetate to arrive at the enzyme activity of the sample. LDH activity measurements were performed in a medium containing 50 mM imidazole/HCl buffer (pH 7.2 at 20°C), 5 mM pyruvate, 0.16 mM NADH, and 100 mM KCl. LDH assay reactions were started by addition of the sample supernatant, and the decrease in absorbance at 340 nm due to NADH oxidation was assayed for total protein content using the Lowry method (Lowry et al., 1951).


RESULTS
CS activity was greater than LDH activity in all species. Charistephane fugiens was the only species with detectable levels of ODH. This illustrates the aerobic nature of ctenophores. No relationship among all species was found between body size (g wet weight) and mass-specific enzyme activity (Fig. 4). However, mass-specific CS activity increased as a function of body size in P. bachei. Both CS and LDH activity increased in nearly all cydippid ctenophores sampled as a function of number of ctenes (Fig. 5).
RELATIONSHIPS BETWEEN CTENE ROWS AND ENZYME ACTIVITIES
Ctenophore body diameter calculated from measured wet weight was used to find number of ctenes. A wet to dry weight conversion factor of 3.95% was used following Hoeger (1983) and Thuesen et al. (2005). Body diameter was calculated from an equation for the relationship between body dry weight and body diameter, W = 2.924D - 1.550, where W=log10 (body dry weight in mg) and D=log10 (body diameter in mm) from Hirota (1972). Based on photographs in Wrobel and Mills (1998) a proportion for number of ctenes to maximum body diameter was found, and the body diameter was calculated from measured wet weight was used to find number of ctenes. The degree of scaling of CS activity to body size in P. bachei (data not shown) was only slightly lower than that found in deep-sea copepods by Thuesen et al. (1998). A stronger relationship exists between number of ctenes and enzymatic activity than between mass and enzymatic activity indicating that amount of metabolic tissue is more important in determining enzymatic activity than body size. LDH activity increases as a function of metabolic tissue size, thus more ctenes could increase an organism's ability to survive hypoxic conditions.
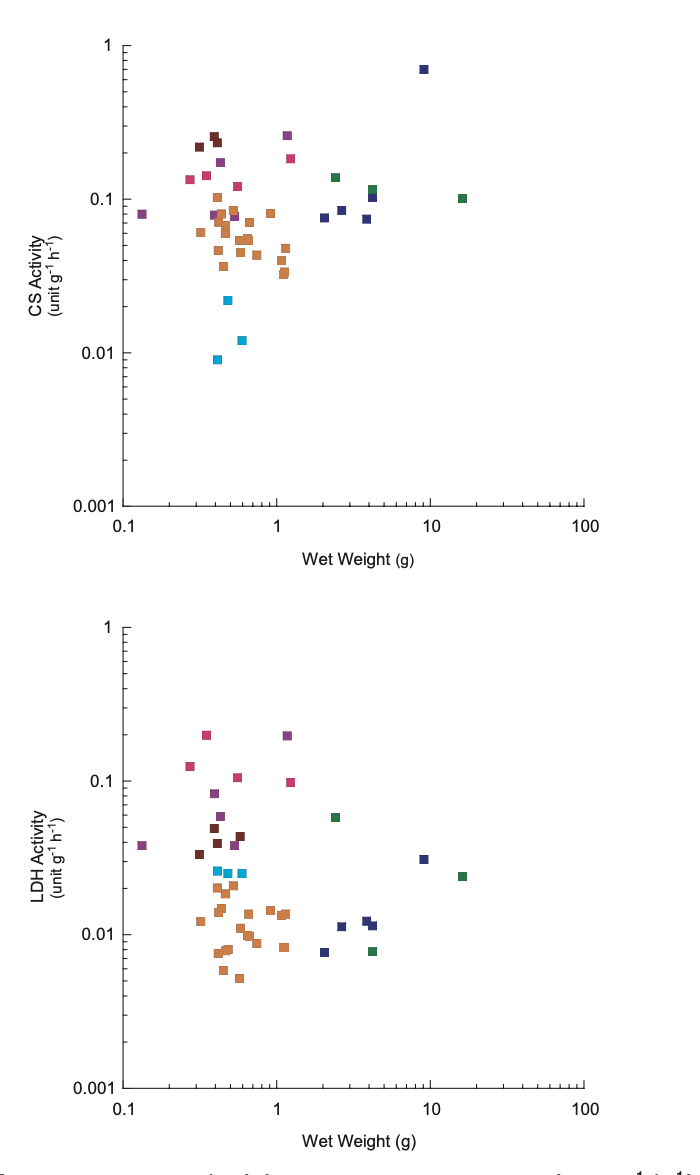
No association was found between mass and either CS or LDH activity. Individual species are depicted by color. Beroe cucumis (dark blue), Beroe forskalii (green), Charistephane fugiens (cyan), Hormiphora californensis (pink), Lampea pancerina (purple), Nephalotena sp. (brown), Pleurobrachia bachei (orange).
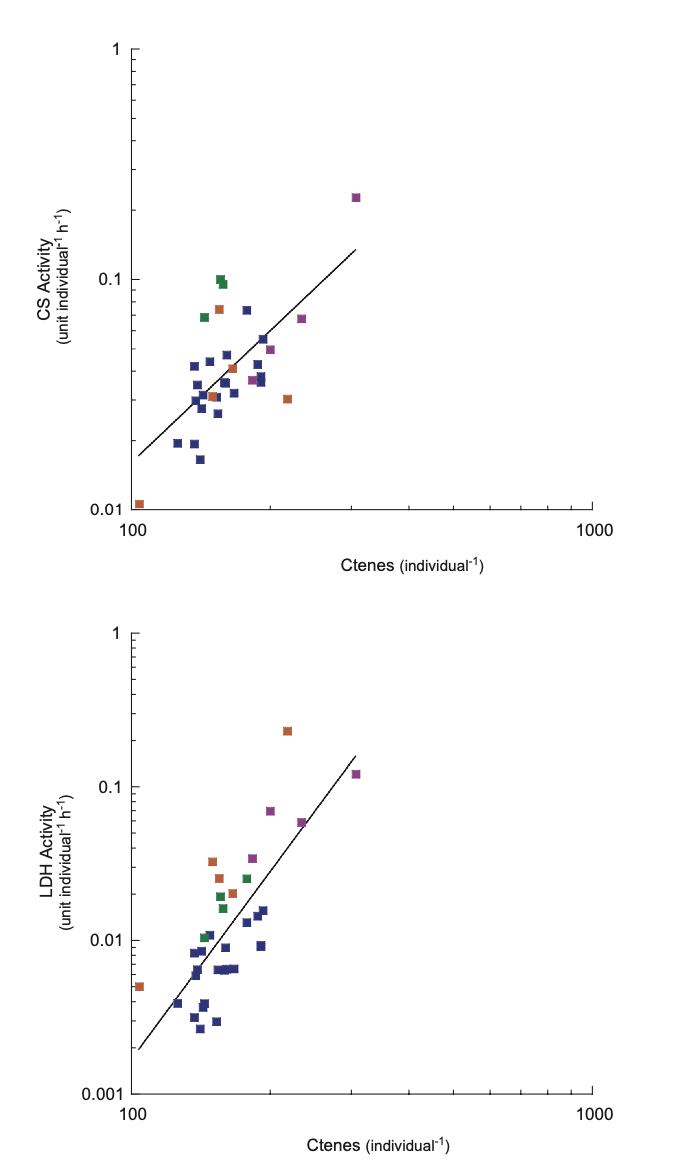
Individual species are depicted by color. Hormiphora californensis (purple), Lampea pancerina (orange), Nephalotena sp. (green), Pleurobrachia bachei (dark blue). With its unusually low number of ctenes, C. fugiens did not seem to fit the same pattern of ctenes to enzyme activity and is not included. The equation of the CS activity regression line is y = 2.54e-6 x 1.90, R2 = 0.566, and the LDH activity regression line is y = 1.28e-11 x 4.06, R2 = 0.351.
CONCLUSIONS
- Ctenophores are mostly aerobic organisms.
- Greater ctene capacity indicates higher CS and LDH.
- Amount of metabolic tissue is more important than body size in determining both aerobic and anaerobic enzymatic activity.
- Ctenophores with higher LDH levels may use anaerobic energy to cope with hypoxic conditions.
PREDICTIONS
In hypoxic estuaries, animal populations are impacted by the lack of oxygen. Environmental hypoxia has been shown to decrease biodiversity and affect estuarine food webs. We predict that organisms with more anaerobic capacity will probably survive the best. Enzyme assays can be used to predict which species will outperform other species as environmental conditions worsen.
LITERATURE CITED
- Childress, J. J. and Somero, G. N., 1979. Depth related enzyme activities in muscle, brain and heart of deep-living pelagic marine teleosts. Marine Biology 52:273-283.
- Diaz, R. J. and Rosenburg, R., 1995. Marine benthic hypoxia - review of ecological effects and behavioral responses on macrofauna. Ann. Rev. Oceanogr. Mar. Bio. 33:245-303.
- Hirota, J., 1972. Laboratory culture and metabolism of the planktonic ctenophore, Pleurobrachia bachei A. Agassiz. In Biological Oceanography of the northern North Pacific Ocean (ed. A. Y. Takenouti), pp. 465-484. Tokyo: Idemitsu Shoten.
- Hoeger, U., 1983. Biochemical composition of ctenophores. Journal of Experimental Marine Biology and Ecology 72:251-261.
- Keister J. E., Houde E. D., Breitburg D. L., 2000. Effects of bottom-layer hypoxia on abundances and depth distributions of organisms on Patuxent River, Chesapeake Bay. Marine Ecology Progress Series 205: 43-59.
- Lowry, O. H., Roseborough, N. J., Farr, A. L., Randall, R. J., 1951. Protein measurement with the folin phenol reagent. Journal of Biological Chemistry 193:265-275.
- Somero, G. N., Childress, J. J., 1980. A violation of the metabolism-scaling paradigm: activities of glycolytic enzymes in muscle increase in larger-size fish. Physiological Zoology 53:322-337.
- Thuesen, E. V., Miller, C. B., Childress, J. J., 1998. Ecophysiological interpretation of oxygen consumption rates and enzymatic activities of deep-sea copepods. Marine Ecology Progress Series 168:95-107.
- Thuesen, E. V., Rutherford Jr., L. D., Brommer, P. L. 2005. The role of aerobic metabolism and intragel oxygen in hypoxia tolerance of three ctenophores: Pleurobrachia bachei, Bolinopsis infundibulum and Mnemiopsis leidyi. Journal of the Marine Biological Association of the United Kingdom 85.
- Wrobel, D. and Mills, C. E., 1998. Pacific Coast Pelagic Invertebrates: A Guide to the Common Gelatinous Animals. Sea Challengers, Monterey, Ca.
ACKNOWLEDGEMENTS
We would like to thank Steven Haddock, Emily Vlahovich, Trisha Towanda, Mario Gadea, Ladd Rutherford, Shane Peterson and Aaron Barnes. This project was funded by an Evergreen Foundation student research award and a Friends of The Evergreen State College Library senior project award.